Columns
Efforts to Standardize Real World Data
What is Real World Data?
Real World Data (RWD) in health care is anonymized data derived from a number of sources that are associated with outcomes in a heterogeneous patient population in real-world settings such as daily clinical practice; RWD includes electronic health records, medical practitioners' receipt for health insurance claim , DPC (Diagnosis Procedure Combination) , and prescription data. More people have undertaken efforts to create usage of RWDs for drug discovery and development on a national scale. In drug development, RWD has already been utilized to visualize unmet medical needs and to optimize designs of clinical trials; in the near future, RWD would be used to serve as historical control in clinical trials. In drug discovery applications, RWD is anticipated to facilitate searching for novel targets and biomarkers for disease. Analyses of RWDs may help identify characteristics of people who may respond to a certain drug; that could lead to discovery of a new drug target.
Utility of Real World Data in health care!!
Data of health insurance claim, DPC data, and disease registry data are currently the main sources of RWD in health care. Electronic medical record data contains more clinical information than other data. However, there are still remaining challenges to utilize it broadly. One of the major challenges is standardization of data.
Electronic medical record system is characterized by the collection of data in various modalities, styles, and formats. There are many different types of data-text data for clinical observations, image data (color or black and white) for X-ray and CT, and waveform data for electrocardiogram chart, etc. These data are intended for in-hospital medical records of daily practice for patients, and are not intended for sharing with other institutions for non-clinical practice purpose.
For example, medical institutions often use their own codes to record drugs, tests, and disease identification. In order to integrate multicenter data for analysis, all institutions are required to use common codes (standardized codes). However, taking medical drugs as an example, usage rate of Standard Master for Pharmaceutical Products (HOT reference code1)), the standard codes of medical drugs provided by the ministry of Health, Labour and Welfare in Japan, is still low in Japan. Therefore, we are developing a system that enables to introduce multiple standard codes by one-touch action. By making it available in a cloud-based environment, we aim to expand standardization of data not only in our hospital but also in hospitals across the country2). If standardization of electronic medical records advances, it will be used not only as a records of daily medical care but also as RWD for research and development for future medical care.
Future of Real World Data
Dr. Kazuhiko Ohe who is a professor of our department set up the "![]() NeXEHRS Consortium3)" that discusses how to operate next-generation health information platforms, and is leading discussions on future health care and data management with participants not only from the medical community but also from private companies with various backgrounds.
NeXEHRS Consortium3)" that discusses how to operate next-generation health information platforms, and is leading discussions on future health care and data management with participants not only from the medical community but also from private companies with various backgrounds.
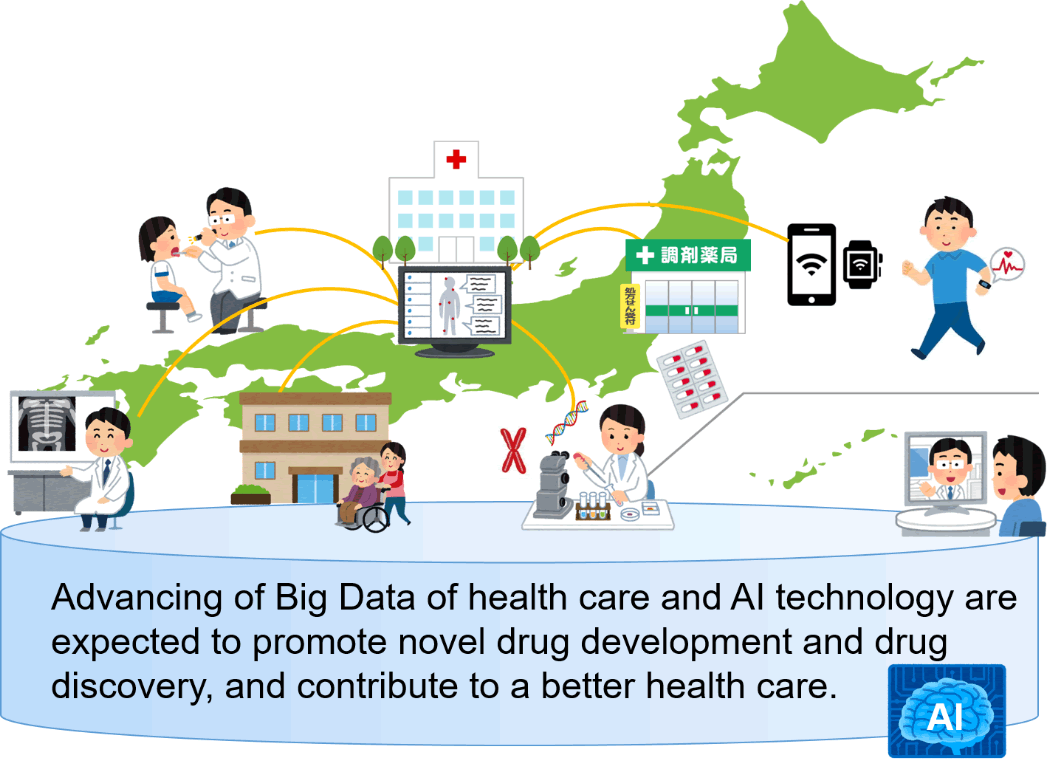
Rapid advancement of IoT and ICT enables accumulation of more detailed personal health records-daily real-time bio-and behavioral information obtained out of hospital-in addition to electronic medical records obtained at hospital. Substantial efforts are made to conduct genomic medicine, and research on linking clinical data with gene mutation information is being actively conducted. Advancing of Big Data of health care and AI technology are expected to promote novel drug development such as the search for biomarkers and surrogate endpoints, and efficient implementation of clinical trials that may lead to new findings and contribute to a better health care.
References
- Promotion of informatization in the medical field https://www.mhlw.go.jp/stf/seisakunitsuite/bunya/kenkou_iryou/iryou/johoka/index.html on the promotion of computerization in the healthcare sector (Japanese only)
- Satomi NAGASHIMA and Kazuhiko OHE. Standard coding systems of pharmaceutical products in Japan and the proposal of integrated coding table, Japan Journal of Medical informatics 39(4): 205-216
- NeXEHRS consortium, https://nexehrs-cpc.jp/
May 2020
Satomi Nagashima
Project Researcher
Department of Healthcare Information Management
University of Tokyo Hospital
| i. | A document describing the details of medical care, examinations, and prescription drugs issued by medical institutions and pharmacies in order to bill public institutions to pay for insurance payments for medical expenditures. |
| ii. | Mechanism of fixed payments based on diagnosis group classification. |
| iii. | A database that manages the patient's illness, treatment details, and treatment course. |




