Columns
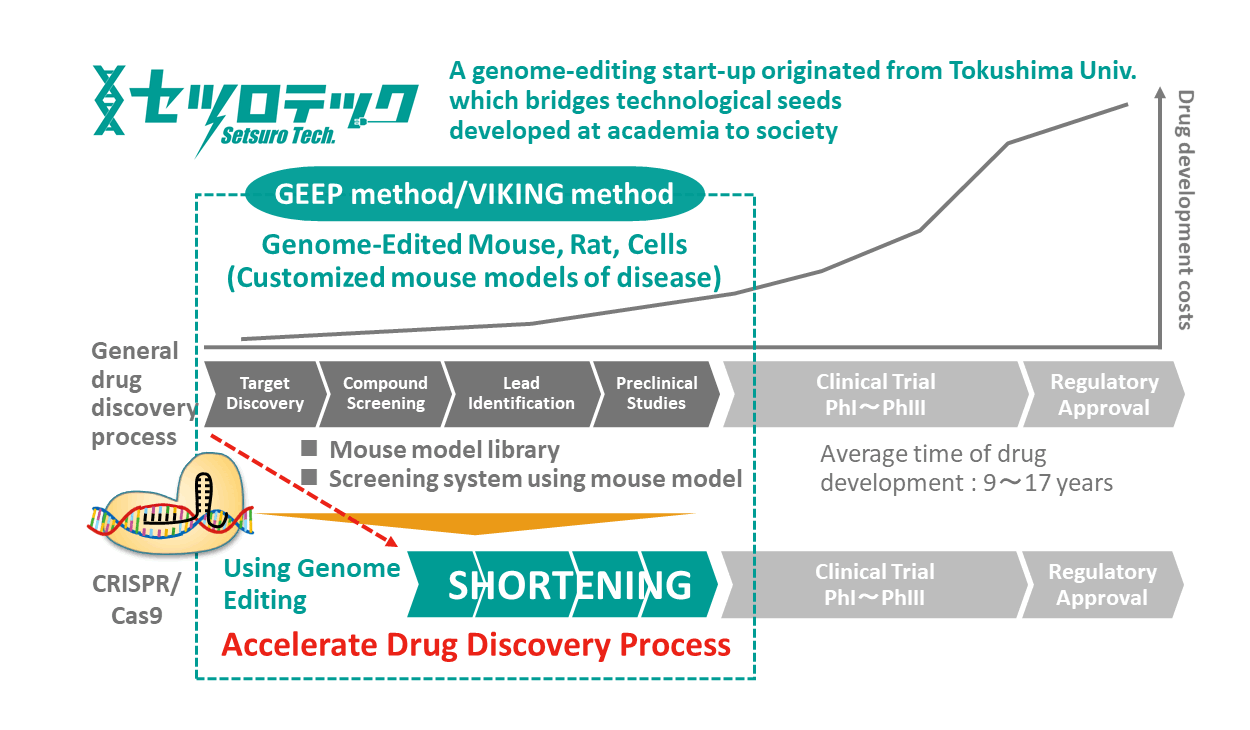
Accelerating Drug Discovery Process with Genome Editing Technology from Tokushima
In a series of drug development processes from basic research to non-clinical and clinical trials, genome editing technologies have been making innovative breakthroughs in every situation. Also, the technology itself has constantly been advancing. In particular, genome editing using CRISPR /Cas9 system, which was awarded the Nobel Prize in Chemistry in 2020, has greatly streamlined the production of knockout mice and disease-model mice, which have been used in a variety of drug discovery research and non-clinical trials. ![]() Setsuro Tech Inc. (Setsurotech) develops new intellectual properties by applying genome editing technological seeds originated in Tokushima University. We aim to take a significant role in establishing a bridge between basic technology and society. With this company vision, Setsurotech supports research and development activities from basic research to practical application.
Setsuro Tech Inc. (Setsurotech) develops new intellectual properties by applying genome editing technological seeds originated in Tokushima University. We aim to take a significant role in establishing a bridge between basic technology and society. With this company vision, Setsurotech supports research and development activities from basic research to practical application.
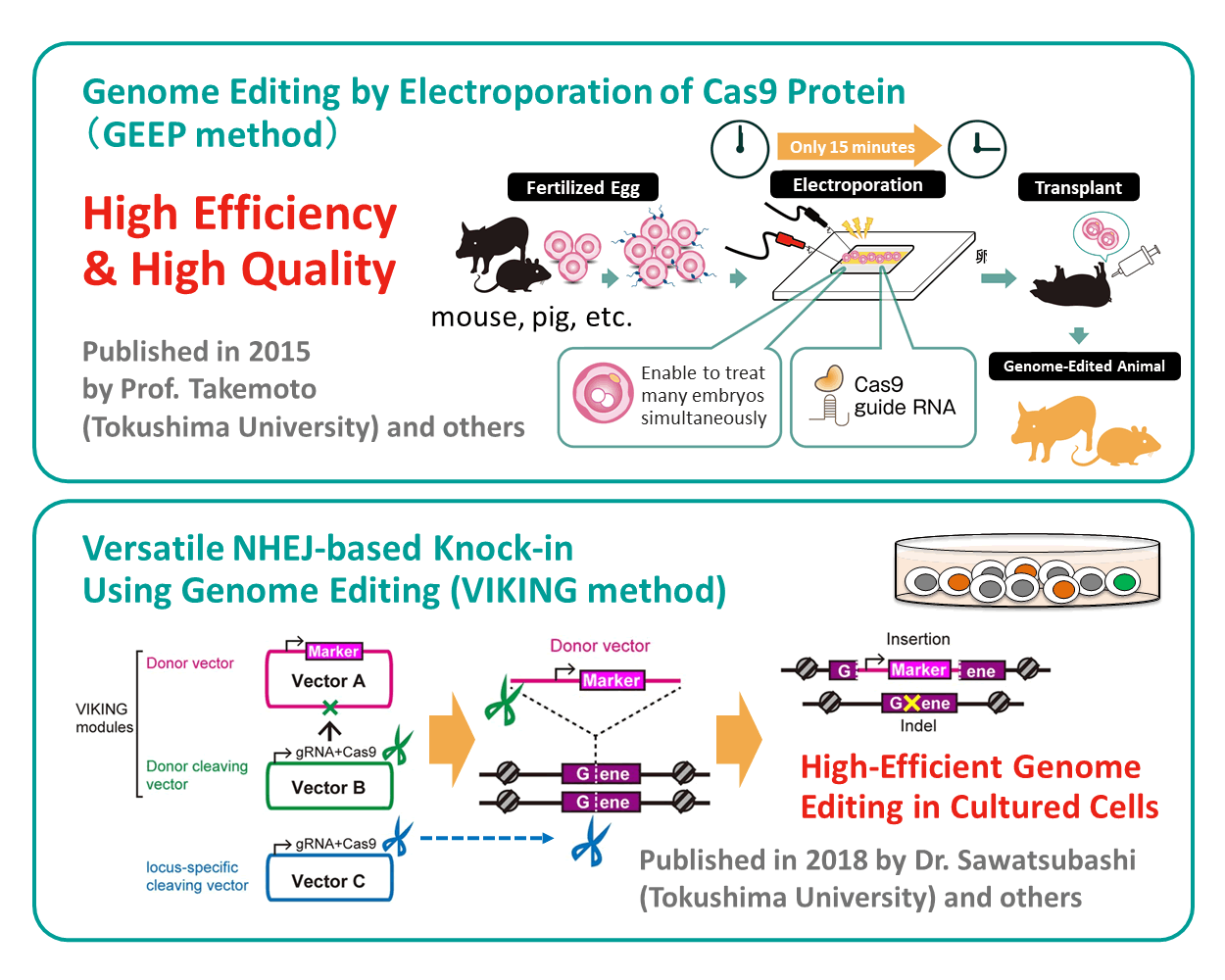
Setsurotech is founded in 2017 by Tatsuya Takemoto and Shun Sawatsubashi, who are also faculty members of Tokushima University. Fundamental technologies of Setsurotech are two genome editing technologies; GEEP method (Genome Editing by Electroporation of Cas9 Protein, WO2017/170771) developed by Tatsuya Takemoto et al. and VIKING method (Versatile NHEJ-based Knock-in Using Genome Editing, JP2018/126122) established by Shun Sawatsubashi et al. Those two technologies enable Setsurotech to do high-throughput genome editing.
GEEP method introduces genome editing factors such as Cas9 protein and gRNA into fertilized eggs by electricity (electroporation)(1-3). Conventionally, the microinjection method has mainly been used to produce genetically modified mice. This method uses a glass capillary, a very thin glass tube, to inject the solution into fertilized eggs one by one under a microscope. Therefore, it requires specialized equipment and advanced embryo manipulation techniques. With the GEEP method, on the other hand, we can do genome editing only by laying out fertilized eggs on electrodes and even bringing genome-editing factors into a lot of fertilized eggs (20 to 200) at once without any complicated work for each fertilized egg. Thus, the GEEP method enables us to introduce genome editing factors at high throughput and low cost. This efficient introduction of genome editing factors brings out the advantage of carrying out less-invasive genome editing on a number of fertilized eggs with an identical condition in a short period. It is expected to generate genome editing organisms in a highly efficient manner. Our Chairman and CTO, Tatsuya Takemoto (also belongs to Tokushima University) and his colleagues reported the use of the GEEP method for phenotypic modification in pig(3), and the GEEP method is expected to be applied to not only mice but also a wide range of animals.
VIKING method is a genome editing method for cultured cells leveraging non-homologous end-joining (NHEJ) repair pathway occurring more frequently than the homologous recombination that had been conventionally used in genome editing on cultured cells(4). In the VIKING method, to omit the cumbersome donor vector construction process, a knock-in module using vectors set(s) was developed, which is used for the introduction of genome editing tools. Therefore, it allows performing knock-in easily and efficiently in cultured cells available for a gene insertion. One of the strengths of the VIKING method is that it can insert vectors larger than 10kbp, which is highly difficult by using viral vectors. Moreover, it makes the selection of edited cells after genome editing easier because it can insert marker sequences such as a drug resistance gene into the genome. Furthermore, the VIKING method realized a dramatic reduction in unintended sequence modifications (random integration) by optimizing the introduction ratio of the three vectors to be used. With the VIKING method,
It is possible to establish a gene-disrupted strain by inserting a donor vector when the target is the gene coding sequence (CDS). Also, it is possible to establish a stable expression strain by knock-in when the target is a safety-harbor locus such as AAVS1. The VIKING method has been successfully applied in various cell types. Setsurotech is dedicated to in-house research activities on not only the VIKING method but also other methods to meet the demands of several bases deletion and a point mutation to which the VIKING method is not very suitable.
Besides providing the production and delivery service of gene-modified mice and cultured cells using the methods introduced above, Setsurotech aims to enrich the lives of people by exploring and establishing the genome editing industry as a fundamental company providing genome editing technology and organisms. Our research support business, especially genome editing mice, rats, and cultured cells manufacturing service, has been used by various researchers for the last four years since the foundation of Setsurotech and well received by such researchers. So far, our services have contributed to the publication of several papers. For example, the research paper of Kazuhito Naka, an associate professor of the Research Institute for Radiation Biology and Medicine, Hiroshima University were published in the British Journal of Sciences, Nature Communications (2020)(5). This paper includes the analysis of gdpd3 knockout mice provided from Setsurotech and focused on chronic myelogenous leukemia (CML) stem cells responsible for relapse after treatment with tyrosine kinase inhibitions (TKIs). Mr. Naka's team found that the regulation of lysophospholipid metabolism by Gdpd3 played a key role in maintaining the viability of CML stem cells. Dr. Naka said that it is possible to further recurrence of suppression in CML patients if he can develop a targeted drug that inhibits Gdpd3 combined with TK1s in the future. This is one of the good examples of the research results that are considered translational research because they are a seed for practical application in the future (for more details, please refer an introductory article on our website(6) ).
As described above, genome editing technologies are powerful tools to embody and validate researchers' unique ideas. Due to the great potential of this technology for commercial use, however, patent disputes are still ongoing in the US, and the situation is getting more complicated. In order to address this situation and provide our service with confidence, we have signed licensing agreements on CRISPR/Cas9 with both Broad Institute and ERS Genomics regarding usage in research supporting purposes in Japan. Our unique genome editing technology enables researchers to provide genome editing models quickly and at a relatively lower cost. From basic research to screening using disease models and even to nonclinical trials using genome-edited mice, Setsurotech supports researchers engaging in drug discovery and development activities.
Reference
- Hashimoto M, Takemoto T. Electroporation enables the efficient mRNA delivery into the mouse zygotes and facilitates CRISPR/Cas9-based genome editing. Sci Rep. 2015; 5:11315.
- Hashimoto M, Yamashita Y, Takemoto T. Electroporation of Cas9 protein/sgRNA into early pronuclear zygotes generates non-mosaic mutants in the mouse. Dev Biol. 2016; 418(1):1-9.
- Tanihara F, Takemoto T, Kitagawa E, et al., Somatic cell reprogramming-free generation of genetically modified pigs. Sci Adv. 2016;2(9):e1600803.
- Sawatsubashi S, Joko Y, Fukumoto S et al., Development of versatile non-homologous end joining-based knock-in module for genome editing. Sci Rep. 2018; 8(1):593.
- Naka K, Ochiai R, Matsubara E, et al., The lysophospholipase D enzyme Gdpd3 is required to maintain chronic myelogenous leukemia stem cells. Nature Communications 2020; 11(1):4681.
- Practical Case Sharing: Elucidation of significance of lysophospholipid metabolism in cancer stem cell using genome-edited knockout mice (H. Naka, Hiroshima University) https://www.setsurotech.com/engnews/case_210531/
June 2021
Shinichiro Takezawa
CEO
Setsuro Tech Inc.